History of Present Illness
As we approach the patient, we illicit more information from the mother in order to think of the differentials besides or in addition to gastroenteritis. Asking about PO and output would reveal valuable information such as the three "P"s: Polydipsia, Polyphagia, and Polyuria. As we know, these are classic signs of a hyperglycemic patient with Type 1 DM. Asking abouf family history would also have revealed that there was a family history of diabetes. Another clue. Walking in we see a thin kid. This is also a clue. Why? Well, if he is a Type 1 diabetic, he lacks insulin which has many functions including the following which are pertinent:
- Controlling cellular uptake of glucose
- Increasing glycogen synthesis from increased serum glucose
- Increased fatty acid synthesis and conversion to triglycerides to be stored as fuel through esterification
- Decreased proteolysis
- Decreased lipolysis
- Decreased gluconeogenesis
- Increased Potassium (K+) uptake in cells
So what does this mean? It means that lack of insulin leads to the opposite of those functions listed above. There is a decreased uptake of glucose needed for cellular respiration (including the Kreb's Cycle) to provide the energy the body needs to function.
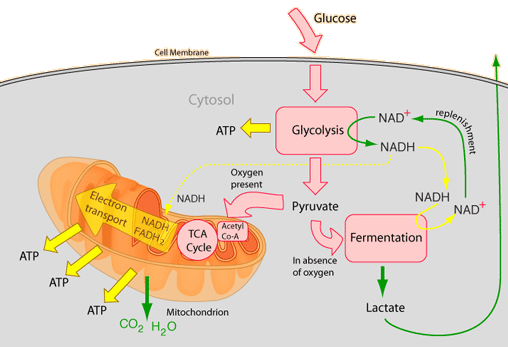 If you are like me, you quickly forget the process of cellular respiration and the Kreb's Cycle. There are quite a few tutorials on line, especially on youtube, if you need some furter education, reeducation, or just more in depth understanding. I advise you to occasionally look what is there.
If you are like me, you quickly forget the process of cellular respiration and the Kreb's Cycle. There are quite a few tutorials on line, especially on youtube, if you need some furter education, reeducation, or just more in depth understanding. I advise you to occasionally look what is there.Okay, so we know that without insulin, the cells are starving in a land of plenty. They cannot gain access to the glucose. Since the cells are starving, and insulin is lacking, we see a breakdown of glycogen in liver and muscle tissue to create more glucose for the cells, but yet, the cells cannot take it in. This leads to a continuing cycle of accumulting glucose. So we do a bedside glucometer reading and find it is "CRITICAL HIGH". A patient in DKA will have a blood glucose > 250 mg/dl, or for those up North or across the pond, >13.8 mmol/l.

Courtesy of http://realitycheck.org/

Courtesy of www.inkeehong.com
It also means an increased mobilization of Free Fatty Acids (FFAs) and a switch of hepatic lipid metabolism to ketogenesis. And what does this mean? The body metabolizes triglycerides (lipolysis) and muscle (gluconeogenesis)for energy. This continuing breakdown of adipocytes, muscle and proteins causes the thin body shape. The body needs to replenish, so the patient progressively gets hungrier, thus the polyphagia. Of course with nausea and vomiting, the child may wish to forget about eating as the child gets sicker.
Ketogenesis
Normally with high serum levels of glucose, acetyl-CoA metabolizes the glucose to make FFAs to then be stored as fat in adipose tissue. But, as we discussed the cells are starving so there is a breakdown of adipose tissue into FFAs to be used as energy by muscle and cells and also to be transferred to the liver bound to albumin to be broken down into acetate, to be broken down further into the ketnone body, acetoacetate. The brain can use ketone bodies as fuel when glucose levels are low. There, and other places low in adipose tissue the ketoacids are oxydized back into acetyl-CoA to be used in cellular respiration. I know this is getting deep, but...well try to hang in there. Acetyl-CoA needs the organic chemical oxaloacetate to push it through the Kreb's Cyle. But oxaloacetate is used up during gluconeogenesis, so there is not enough to process the acetyl-CoA, which means the excess goes back to ketone body formation. Here is the reaction:
Acetyl CoA < > Acetoacetic Acid (acetoacetate) < > Beta-hydroxhybutyric Acid
This leads to ketosis, the build-up of excess ketones. Not usually a bad thing as it is can be common in fasting or ketogenic diet, but this time it is a bad thing.

Along with glucose, the kidneys try to excrete the excess ketones and on a urine dipstick, ketones >3+ indicate ketoacidosis. Urine dipstick? They are commonly done in the ED. Since the patient has polyurea, collecting a urine sample is quick and it only takes a minute to dip the urine stick and read it. I foresee, if our roles in EMS increase, we may have the option of using these in 10 years. Far fetched I am sure, but if we take advantage of the patient's need to urinate, we can learn many things from the dipstick such as not only determining DKA by identifying large quantaties of ketones and glucose in the urine, but recognizing kidney stones and UTIs as well.
Courtesy of www.medicalgeek.com
Acetone
I mentioned a smell when we walk in the room similar to that of nail polish. This is acetone. While acetone smell is a highly reliable sign of ketosis, not everyone has the gene to smell it on someone's breath. It is estimated that only about 30-50% of people have the gene to smell it, but if it is smelled, it is a significant sign. What causes that "fruity odor"?
 Courtesy of http://www.invelos.com
Courtesy of http://www.invelos.comSo as I mentioned above, when the acetoacetate that cannot be metabolized back into Acetyl-CoA or used as fuel are then broken down further into Beta-hydroxybutyrate, a ketoacid like ketoacetate, and acetone as a byproduct. Acetone, as a byproduct cannot be synthesized back to acetyl-CoA. No problem. It is easily excreted in the urine. Acetone also has a high vapor pressure meaning it is easily evaporated through exahalation. Alsa, the infamous "fruity" or acetone odor. This odor is very specific to identifying ketosis. If you are like me in that you cannot smell it, don't feel to depressed. Though I have seen and heard varying numbers, the consensus seems to be more than half the population does not have the gene to smell it. But if you do, and you smell it, then consider it a very important indication of ketoacidosis.
Acidosis
If you are weak on acid base basics, here is a basic overview:
Courtesy of http://www.mountnittany.org/

Our patient is very weak in bed. Why is he weak? A couple of reasons. He is dehydrated and lacking energy since the the cells are having a tough time getting at the glucose they need. There may be a build-up of lactate which fatigues muscle cells. But not only that, Acetoacetate and betahydroxybuterate are ketoacids. They are very acidic and with enough ketoacid accumulation, the pH drops to dangerously low levels, leading to the state of Ketoacidosis. Acidic states hinder enzymatic reactions, and this includes skeletal muscle contractions. The acidotic state leads to the GI symptoms such as the abdominal discomfort, nausea and vomiting, though the exact etiology of these are unknown. Part of the diagnostic criteria for DKA is an arterial pH<7.30.> Now the body will try to buffer this acidotic state, and one way is Kussmaul breathing. Actually, a peson will develop tachypnea or hyperventilation first,and then the Kussmaul breathing at later stages. Hyperventilation and Kussmaul breathing are not necessarily the same. Kussmaul breathing is more like deep, fast and labored breathing and is a later sign and can lead to respiratory fatigue. Still, you will see hyperventilation of some sort, like we saw in our sick kid. Why? To blow off CO2 of course. Most of us know that but for those providers who are not sure of the relevance of "blowing off CO2", let me explain briefly. An acidic environment in the blood means more excess H+ ions. To counter this acidosis, the H+ ions need rid of. The body has many buffering systems to keep the pH between 7.35-7.45, but we will focus on the following equation:
We see when H+ ions accumulate after being donated by the ketone bodies, to become hydronium H3O, they are buffered by HCO3 (bicarbonate, a.k.a bicarb), which binds to the H+ to become H2CO3 (carbonic acid) and H2O, then to be broken down to CO2 and H2O, both of which are exhaled. The increases in H+ ions stimulate central chemoreceptors (tha aortic bodies in the aortic arch and carotid bodies in the carotitids) which in turn stimulate the respiratory centers in the medulla and pons in the brain stem to increase rate and depth of respirations. So the more the body hyperventilates to compensate, the more O2 is inhaled to eventually bind with H+ to make H3O and the more H2O and CO2 is exhaled, trying to raise the pH or keep it level. Remember, a low pH state is actually acidotic. So thus, the reason for the hyperventilation and Kussmal breathing. Usually we start to see the Kussmal breathing when the patient is starting to fatigue from the hyperventilation.
Courtesy of www.Jumpstarttriage.com
What is interesting is the HCO3 is used up in the buffer system- one for each H+ buffered so serum labs and ABGs will show a decrease in HCO3 as well as a normal or decrease in the CO2. This is due to the compensatory mechanism via buffers and hyperventilation. But you knew that. A bicarbonate level of 18 mEq per L or less is part of the diagnostic criteria of DKA, but again, we in the field usually don't have access to serum labs. Another one of the blood tests done in the ER will be an Anion Gap, wich in DKA, will be >12. It is used to help diagnose metabolic acidosis.
Now if you put the kid on the monitor, you may see peaked "T" waves. Why? Insulin pushes K+ back into cells, but there is no insulin, remember. Not only that, but the acidosis pulls the K+ out of the cells. This leads to hyperkalemia, which gives us the peaked "T" waves on the EKG. But remember, the, body's cells don't have the needed K+ as it is in the vacular space. Making matters worse, along with the glucose and ketones, the patient will excrete the K+ and the Na+, leading to hypokalemia and hyponatremia later, especially after fluids are started. But we will discuss that later.
Courtesy of www.learnTheHeart.com
Another EKG finding that though not common, but occasionally occurs, is a prolonged QTc. You should know about prolonged QT from Adam in Long QT Part 1 Part 2, Part 3. This is a reason these kids need cardiac monitoring.Just remember that hyperkalemia is in no way diagnostic criteria but something you may see before rehydration and insulin therapy. Afterward you may see hypokalemia (hopefully not), such as when you may be tasked with doing an interfacility transfer of this kid.
Courtesy http://www.medinfo.ufl.edu/
Again, this is not something to expect to see and not used for diagnostic criteria but just another reason to monitor your pediatric DKA patient.
So, to rehash, I just went over some of the obvious signs we will see and why we see them. Basically what is going that is reflected in the signs and symptoms. I understand for many of you this may have been a little deep, and quite boring. However, maybe not as boring as this guy (who is thorough though).
Referrences:
Barbour, L.A., Friedman, J.E., Chapter 26, "Management of Diabetes in Pregnancy", Influence of Metabolic Changes in Pregnancy on Diabetes Management. March 6, 2003: http://www.endotext.org/diabetes/diabetes36/diabetesframe36.htm
Brandis, Kerry. "8.2.1. What is ketoacidosis?, Acid-base pHysiology", Anaesthesia Education Website<>, http://www.anaesthesiamcq.com/AcidBaseBook/ab8_2.php
Chansky Michael E, Lubkin Cary L, "Chapter 211. Diabetic Ketoacidosis" (Chapter). Tintinalli JE, Kelen GD, Stapczynski JS, Ma OJ, Cline DM: Tintinalli's Emergency Medicine: A Comprehensive Study Guide, 6e:
http://www.accessmedicine.com/content.aspx?aID=606036
Crandall, Jill P. MD, "Diabetic Ketoacidosis (DKA)", The Merck Manuals Online Medical Library, May 2007, http://www.merck.com/mmpe/sec12/ch158/ch158c.html
"Diabetic Ketoacidosis Pathophysiology", slide presentation, The Diabetes Monitor: http://www.diabetesmonitor.com/dmemerh/tsld005.htm
Faulke, Dan. DKA Mechanisms Anaesthetist.com June 2008
http://www.anaesthetist.com/icu/organs/endocr/dm/Findex.htm#dka.htm
Gundstream, Stanley, E., Anatomy and Physiology: With Integrated Study Guide, 4th Ed., McGraw Hill, 2010
Klabunde, Richard, E., “Chemoreceptors”, Cardiovascular Physiology Concepts, http://www.cvphysiology.com/, 04/01/07
Kuppermann, N, Park, J, Glatter, K, Marcin, J, Glaser, N. Prolonged QT Interval Corrected for Heart Rate During Diabetic Ketoacidosis in Children,
ARCH PEDIATR ADOLESC MED/VOL 162 (NO. 6), JUNE 2008
Downloaded from http://archpedi.ama-assn.org/ at University of Pittsburgh, on October 20, 2009
Musa-Veloso, K, Likhodii, S., Cunnane, S. "Breath acetone is a reliable indicator of ketosis in adults consuming ketogenic meals", American Journal of Clinical Nutrition, Vol. 76, No. 1, 65-70, July 2002: http://www.ajcn.org/cgi/content/full/76/1/65
Patlak, Joe, "ISF-Plasma Exchange/Capillary Pressures", Body Fluids lectures of Medical Physiology 301, http://physioweb.med.uvm.ed%2011/01/00
Place Frederick, Mayer Thom, "Chapter 128. Diabetic Ketoacidosis" (Chapter). Tintinalli JE, Kelen GD, Stapczynski JS, Ma OJ, Cline DM: Tintinalli's Emergency Medicine: A Comprehensive Study Guide, 6e: http://www.accessmedicine.com/content.aspx?aID=597225
Sateesh, B, "Glucose Homeostasis and Diabetes", Pharmainfo.net, Vol. 5 Issue 4, 08/09/2007: http://www.pharmainfo.net/reviews/glucose-homeostasis-and-diabetes







2 comments:
Brilliant post - I learnt a lot from that that I didn't even know I didn't know before reading it.
That is my goal. Believe it or not, I was a medic a long time before I learned the pathophysiology at this level. I could have gone a little deeper at a molecular level but I that would mean reliving the horror of my Organic Chemistry classes I took one summer. Besides, O-chem was not one of my strong points. Either way, I think EMS provider should know the physiology of disease states they are treating.
Post a Comment